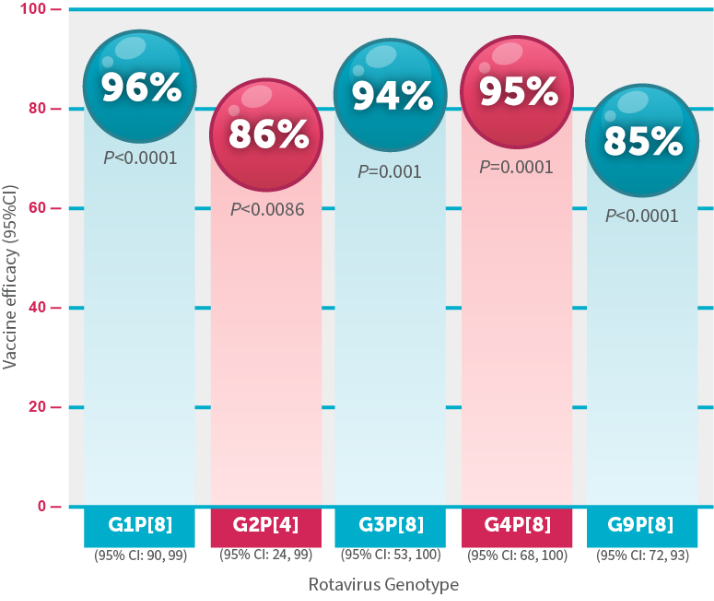
ROTARIX is a vaccine indicated for the prevention of rotavirus gastroenteritis caused by G1 and non-G1 types (G3, G4, and G9) when administered as a 2-dose series. ROTARIX is approved for use in infants 6 weeks and up to 24 weeks of age.
ROTARIX is a vaccine indicated for the prevention of rotavirus gastroenteritis caused by G1 and non-G1 types (G3, G4, and G9) when administered as a 2-dose series. ROTARIX is approved for use in infants 6 weeks and up to 24 weeks of age.
ROTARIX is a vaccine indicated for the prevention of rotavirus gastroenteritis caused by G1 and non-G1 types (G3, G4, and G9) when administered as a 2-dose series. ROTARIX is approved for use in infants 6 weeks and up to 24 weeks of age.
- Contraindications for ROTARIX include a history of any of the following: hypersensitivity to the vaccine or any component of the vaccine; uncorrected congenital malformation of the gastrointestinal tract (such as Meckel’s diverticulum) that would predispose the infant to intussusception; intussusception (in postmarketing experience, intussusception resulting in death following a second dose has been reported following a history of intussusception after the first dose); or Severe Combined Immunodeficiency Disease (SCID)
- Contraindications for ROTARIX include a history of any of the following: hypersensitivity to the vaccine or any component of the vaccine; uncorrected congenital malformation of the gastrointestinal tract (such as Meckel’s diverticulum) that would predispose the infant to intussusception; intussusception (in postmarketing experience, intussusception resulting in death following a second dose has been reported following a history of intussusception after the first dose); or Severe Combined Immunodeficiency Disease (SCID)
- Contraindications for ROTARIX include a history of any of the following: hypersensitivity to the vaccine or any component of the vaccine; uncorrected congenital malformation of the gastrointestinal tract (such as Meckel’s diverticulum) that would predispose the infant to intussusception; intussusception (in postmarketing experience, intussusception resulting in death following a second dose has been reported following a history of intussusception after the first dose); or Severe Combined Immunodeficiency Disease (SCID)
- The tip caps of the prefilled oral dosing applicators contain natural rubber latex, which may cause allergic reactions
- Administration in infants suffering from acute diarrhea or vomiting should be delayed. Safety and effectiveness in infants with chronic gastrointestinal disorders have not been evaluated
- Safety and effectiveness in infants with known primary or secondary immunodeficiencies, including infants with human immunodeficiency virus (HIV), infants on immunosuppressive therapy, or infants with malignant neoplasms affecting the bone marrow or lymphatic system have not been established
- Rotavirus shedding in stool occurs after vaccination with peak excretion occurring around Day 7 after Dose 1. One clinical trial demonstrated that vaccinees transmit vaccine virus to healthy seronegative contacts. Caution is advised when considering whether to administer ROTARIX to individuals with immunodeficient close contacts
- A study conducted after approval of ROTARIX showed an increased risk of intussusception in the 31 days after the first dose of ROTARIX, especially in the first 7 days. In other reports, an increased risk of intussusception has been observed within 7 days after dose 2
- Safety and effectiveness of ROTARIX when administered after exposure to rotavirus have not been evaluated
- In clinical studies, common (≥5%) solicited adverse reactions among recipients of ROTARIX included fussiness/irritability, cough/runny nose, fever, loss of appetite, and vomiting
- Safety and effectiveness of ROTARIX in infants younger than 6 weeks or older than 24 weeks of age have not been evaluated
- The effectiveness of ROTARIX in pre-term infants has not been established
- Vaccination with ROTARIX may not result in protection in all vaccine recipients
Please see full Prescribing Information for ROTARIX.